Europium points to new suspect in continental mystery
Study: Rare earth element implicates garnet for continents’ missing iron
HOUSTON — (May 16, 2018) — Clues from some unusual Arizona rocks pointed Rice University scientists toward a discovery — a subtle chemical signature in rocks the world over — that could answer a long-standing mystery: What stole the iron from Earth’s continents?
The find has weighty implications. If the iron content of continental rocks was a bit greater, as it is in the rocks beneath Earth’s oceans, for example, our atmosphere might look more like that of Mars, a planet so littered with rusty, oxidized rocks that it appears red even from Earth.

From left, Rice University’s Cin-Ty Lee, Ming Tang and Graham Eldridge with one of the Arizona xenoliths that prompted their find. (Photo by Jeff Fitlow/Rice University)
In a new paper available online in Science Advances, Rice petrologists Cin-Ty Lee, Ming Tang, Monica Erdman and Graham Eldridge make a case that garnet steals the most iron from continents. The hypothesis flies in the face of 40-plus years of geophysical thinking, and Tang, a postdoctoral fellow, and Lee, professor and chair of the Department of Earth, Environmental and Planetary Sciences at Rice, said they expect a healthy dose of skepticism from peers.
“The standard view, which even we agreed with and wrote papers agreeing with, is that iron is removed from continental crust by another mineral called magnetite,” Lee said. “I think people haven’t thought much about garnet, possibly because it doesn’t show up very much and magnetite shows up in a lot of samples.”
Building a case for or against either mineral isn’t easy because the iron they’re accused of stealing disappears many miles below active volcanoes. The prime example today is the arc of volcanoes that span the Andes Mountains in South America. Similar continental arcs are believed to have formed much of Earth’s major landmasses, but scientists have no instruments capable of directly observing what happens beneath continental volcanic arcs. Instead, the missing iron mystery must be solved with deductive reasoning about Earth’s inner workings and rare rocks that hold clues from the scene of the crime.

The Cordillera Huayhuash in the Peruvian Andes as seen from the International Space Station in May 2008. (Image courtesy of ISS Expedition 17/NASA)
“The accepted wisdom is that magnetite pulls iron from the melt before the melt rises and gets erupted out at continental arcs,” Tang said. “Iron depletion is most pronounced at continental arcs, where the crust is thick, and much less so in island arcs, where the crust is thin. However, there is no obvious explanation for why the extent of magnetite involvement would correlate with thickness of the crust.”
But garnet does correlate. Almandine, an iron-laden type of garnet, is more easily made under high pressure and high temperature — the kind of conditions that exist in the subduction zone beneath the Andes, where continental crust can be as much as 50 miles thick, Lee said.
Tang might never have suspected garnet were it not for a field trip by Lee and students to central Arizona in 2009 to look for xenoliths.
“‘Xeno’ meaning foreign and ‘lith’ meaning rock,” Lee said. “They are much older than the volcanoes they came from. These volcanoes ripped up the rocks from 60 to 80 kilometers deep, and the xenoliths came up as little fragments. It’s difficult to find rocks like this, but when you do, they give you a window, a direct window, into the deep parts of the continental arc, the root.”
Erdman, then a doctoral student in Lee’s lab, conducted an initial analysis of the xenoliths, and established that they were formed in a continental arc setting and were rich in garnet. Two years later, Rice undergraduate Graham Eldridge spent a summer characterizing rare earth elements in the xenoliths and found hints that they contained unusual Europium ratios.
Europium typically forms minerals that allow each of its atoms to share three electrons with nearby atoms, an “oxidation state” that chemists notate as Eu+3. Europium also forms minerals in which it shares two electrons, and the notation for this less-oxidized state is Eu+2. In an oxygen-plentiful environment, Europium occurs in its highest oxidation state (Eu+3), but at more intermediate levels of oxygen in the mantle it can occur in both Eu+2 and Eu+3 states.
The oxidation states of the Europium that Eldridge found in the Arizona xenoliths suggested they formed in less-oxidized conditions than would be expected in the magnetite scenario, but there was not enough data to confirm this hunch.
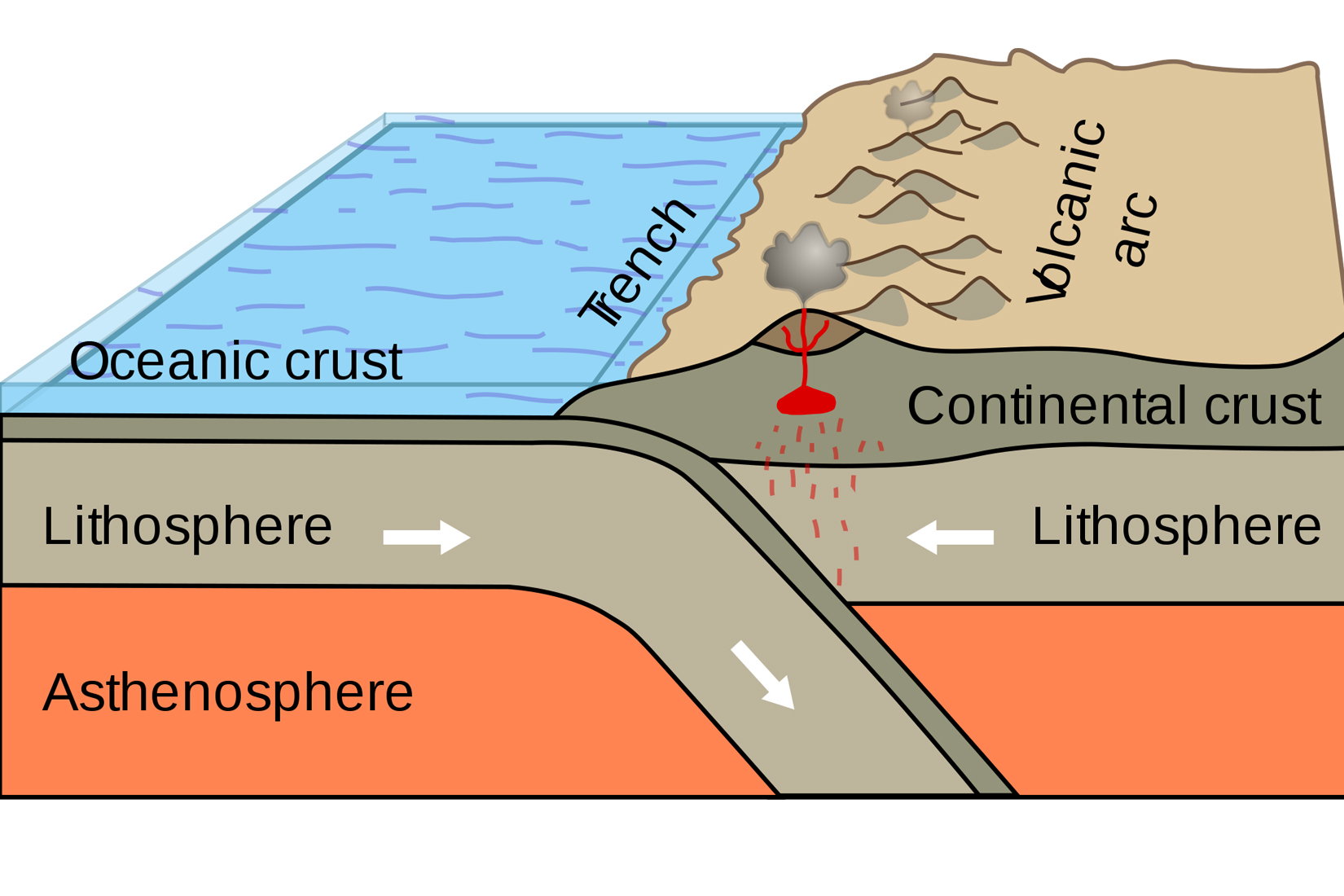
A schematic illustration of plate subduction beneath a continental arc. (Image courtesy of Booyabazooka/Wikimedia Commons)
“Continental arcs happen at subduction zones, where an oceanic tectonic plate slides below a continental plate,” Lee said. “When the oceanic plate is recycled back into the mantle, it is widely thought to introduce a lot of oxygen into the mantle. The magnetite scenario for iron depletion relies heavily on the idea that these subduction zones are highly oxidized at depth.”
Tang joined Lee’s group in 2016 and was intrigued by the Europium ratios in the xenoliths. Tang had extensive experience characterizing Europium as part of his doctoral studies at the University of Maryland, and he began conducting hundreds of painstaking measurements to more precisely characterize the Europium ratios in the Arizona xenoliths.
The quality of Tang’s data not only confirmed the low-oxidation Europium ratios but allowed him to develop a new hypothesis that tied everything together: the garnet, the Europium ratios and the fact that thicker continental crusts are more iron-depleted than thinner island arc crusts.
“As the crustal column gets thicker and thicker, as it is at continental arcs, the temperature and pressure are great enough to produce these iron-rich garnets, which are heavy and sink out,” Tang said. “The iron they pull out is ferrous iron (Fe+2) and not highly oxidized. It goes back into the mantle, and the iron that remains in the melt and erupts to become part of the continental crust becomes even more oxidized on its way to the surface.”
To test the hypothesis on a global scale, Tang spent several months examining records in the Max Planck Institute’s GEOROC database, a comprehensive global collection of published analyses of volcanic rocks and mantle xenoliths collected all over the world.

A garnet pyroxenite xenolith from Sierra Nevada, Calif. (Image courtesy of C. Lee/Rice University)
“There is a relationship between iron depletion and the garnet fractionation signatures, which means magmas that fractionate more garnet are more depleted in iron,” Tang said. “This is born out in the global record, but the evidence is something that wouldn’t be obvious from looking at just one or two cases. It’s the kind of thing that requires a global database, and those have only recently become available.”
Lee said the find has important implications for Earth’s ability to sustain an oxygen-rich atmosphere.
“Photosynthesis produces oxygen, but the primary thing that takes oxygen out of circulation for a long time is oxidation with the crust,” Lee said. “If what comes out of volcanoes to form the continents is effectively already rusted, then it won’t immediately react with and deplete the oxygen in the atmosphere.”
After submitting their results for peer-reviewed publication, Tang and Lee found that renowned Australian petrologist Ted Ringwood and colleagues had implicated garnet rather than magnetite in a few papers published 50 years ago.
“Many of the people in our field have a scientific lineage that goes back to Ringwood,” Lee said. “I’m sure many of them may take one look and think this is a crazy idea, but considering that their great-great-grandfather, academically speaking, had speculated on this, perhaps we’re in good company.”
The research was supported by a Frontiers of Earth Systems and Dynamics grant from the National Science Foundation.
High-resolution IMAGES are available for download at:
https://eol.jsc.nasa.gov/SearchPhotos/photo.pl?mission=ISS017&roll=E&frame=7322
CAPTION: The Cordillera Huayhuash in the Peruvian Andes as seen from the International Space Station in May 2008. (Image courtesy of ISS Expedition 17/NASA)
http://news.rice.edu/files/2018/05/0516_GARNET-grp6-lg-1grbscr.jpg
CAPTION: From left, Rice University’s Cin-Ty Lee, Ming Tang and Graham Eldridge with one of the Arizona xenoliths that prompted their find. (Photo by Jeff Fitlow/Rice University)
http://news.rice.edu/files/2018/05/0516_GARNET-pyroxenite-lg-1e6yvg4.jpg
CAPTION: A garnet pyroxenite xenolith from Sierra Nevada, Calif. (Image courtesy of C. Lee/Rice University)
https://commons.wikimedia.org/wiki/File:Active_Margin.svg
CAPTION: A schematic illustration of plate subduction beneath a continental arc. (Image courtesy of Booyabazooka/Wikimedia Commons)
The DOI of the Science Advances paper is: 10.1126/sciadv.aar4444
A copy of the paper is available at: http://advances.sciencemag.org/content/4/5/eaar4444

